India’s Remarkable Milestone in Cancer Treatment: CAR-T Cell Therapy Triumph India witnesses the success of its indigenous CAR-T cell therapy as the first patient, Dr (Col) VK Gupta, achieves cancer remission.
This innovative cancer treatment, recently greenlit for commercial use by the Central Drugs Standard Control Organisation (CDSCO), involves genetically reprogramming a patient’s immune system to combat cancer cells effectively.
**Understanding CAR-T Cell Therapy**
CAR-T cell therapy, a cutting-edge form of immunotherapy, entails modifying a patient’s T cells in the laboratory to express a specialized protein known as a chimeric antigen receptor (CAR). This CAR protein equips the T cells to recognize and target cancer cells when reintroduced into the patient’s body.
**India’s Indigenous Triumph: NexCAR19**
In October 2023, CDSCO granted approval for NexCAR19, the first locally developed CAR-T cell therapy. Developed by ImmunoACT, a company incubated at the esteemed Indian Institute of Technology Bombay (IITB) and Tata Memorial Hospital, NexCAR19 is designed specifically for treating B-cell cancers, including leukemia and lymphoma.
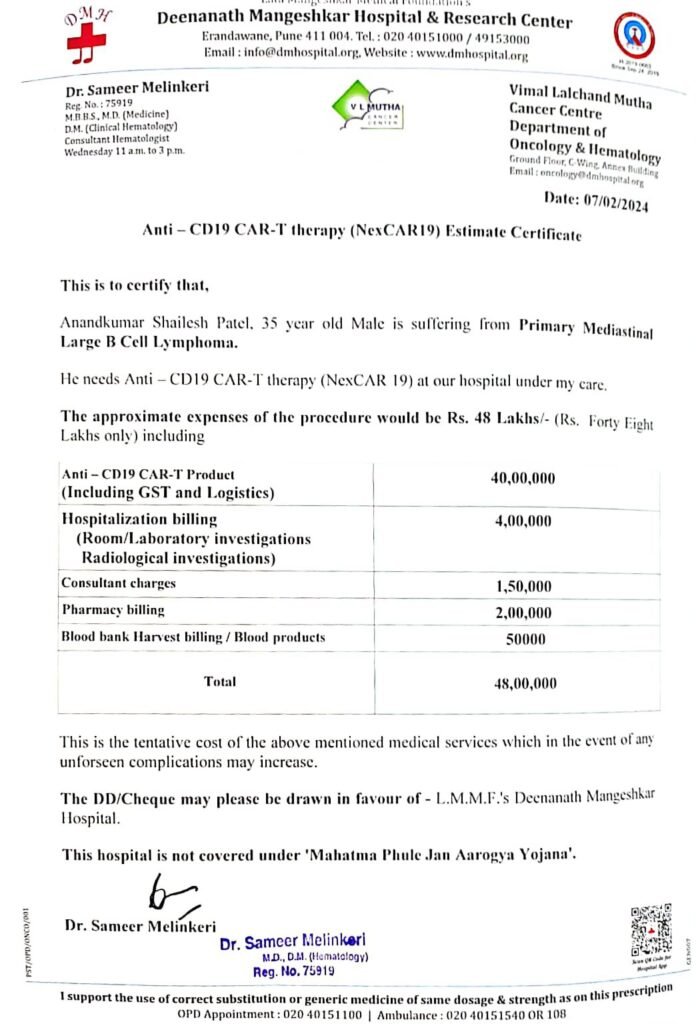
The success story of Dr (Col) VK Gupta highlights the accessibility of this revolutionary therapy in India. Notably, the cost of treatment for Dr Gupta was significantly lower at Rs 42 lakh compared to the Rs 4 crore expenses typically incurred abroad.
Here is a photocopy of patient bill
**Expanding Reach Across India**
NexCAR19 is currently available in over 30 hospitals across more than 10 cities in India. Eligible patients, aged 15 and above, suffering from B-cell cancers can now access this cutting-edge treatment.
This achievement underscores the significance of indigenous medical advancements in India’s battle against cancer. For more information on CAR-T cell therapy and its impact on cancer treatment, explore the [official website of ImmunoACT](https://www.immunoact.com/) and the [CDSCO approval announcement](https://cdsco.gov.in/opencms/opencms/en/Home/). Witness the transformative power of CAR-T cell therapy in shaping the future of cancer treatment in India.